Question
- Leaving Cert. Chemistry (Higher) 2015: Section B Q8
- Back to the question >
Answer
(a)
A: Propene
B: Propan-2-ol
C: Propanone
(b)
Polypropene
(c)
Bonds broken:
6 C-H
2 C-C
1 C=O
Bonds formed:
7 C-H
2 C-C
1 C-O
1 O-H
(d)
Isomer: Propanol
Aldehyde: Propanal
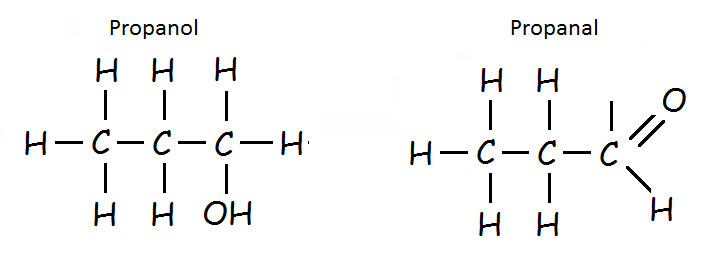
The aldehyde can be converted to alcohol by hydrogenation, using hydrogen passed over the surface of a heated catalyst such as nickel.
(e)
Compound A:
Propene = -48o. This has the lowest boiling point because there are weak dipole-dipole interaction between the molecules, so they are easy to separate.
Compound B:
Propanol = 82o. This has a hydroxyl group, so the intermolecular bonding between the molecules is hydrogen bonding, which is the strongest intermolecular force; hence, this has the highest boiling point.
Compound C:
Propanone = 56o. This contains a carbonyl group, so there are stronger intermolecular forces between these polar molecules; hence, the boiling point is higher.
A: Propene
B: Propan-2-ol
C: Propanone
(b)
Polypropene
(c)
Bonds broken:
6 C-H
2 C-C
1 C=O
Bonds formed:
7 C-H
2 C-C
1 C-O
1 O-H
(d)
Isomer: Propanol
Aldehyde: Propanal
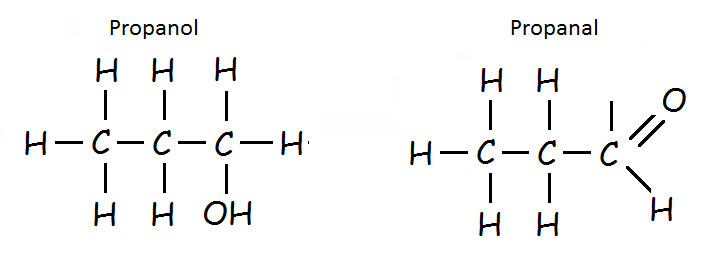
The aldehyde can be converted to alcohol by hydrogenation, using hydrogen passed over the surface of a heated catalyst such as nickel.
(e)
Compound A:
Propene = -48o. This has the lowest boiling point because there are weak dipole-dipole interaction between the molecules, so they are easy to separate.
Compound B:
Propanol = 82o. This has a hydroxyl group, so the intermolecular bonding between the molecules is hydrogen bonding, which is the strongest intermolecular force; hence, this has the highest boiling point.
Compound C:
Propanone = 56o. This contains a carbonyl group, so there are stronger intermolecular forces between these polar molecules; hence, the boiling point is higher.
