Question
- Leaving Cert. Chemistry (Higher) 2016: Section B Q8
- Back to the question >
Answer
(a)
A: Ethanol
Polymer B: Polyethene
(b)
X: Aluminium oxide
Organic reaction type: Elimination/Dehydration reaction
In ethanol the carbons have a tetrahedral geometry but in ethene they have a planar geometry.
(c)
Organic reaction type: Ethane to chloroethane is a substitution reaction.
Mechanism for substitution
Step1: Initiation
The UV light splits the chlorine molecule into two chlorine atoms.
Step 2: Propagation
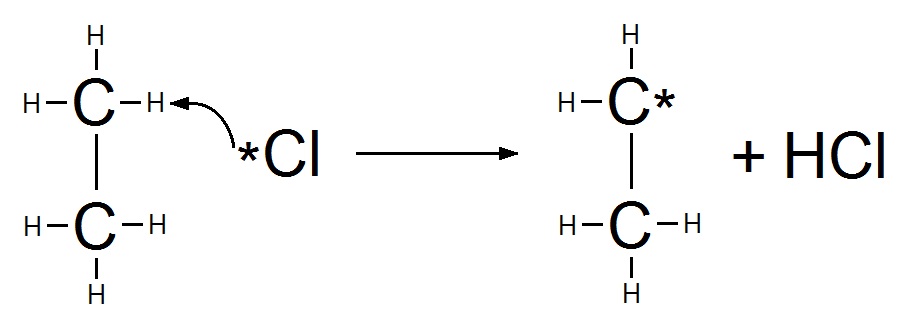
One chlorine atom attacks the ethane molecule removing a hydrogen, forming hydrogen chloride and an ethyl free radical.
Step 3: Termination
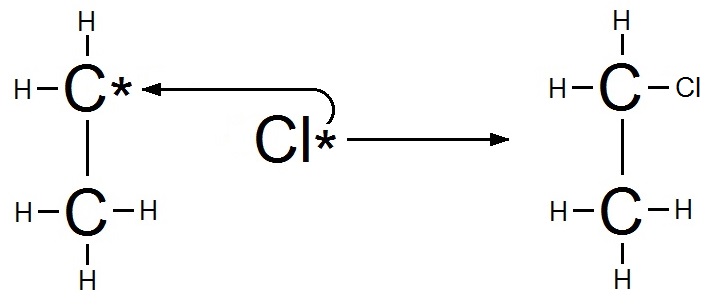
The other chlorine atom then combines with the free radical to form chloroethane.
The addition of tetraethyllead will speed up the reaction.
(d)
C: Ethylmethanoate
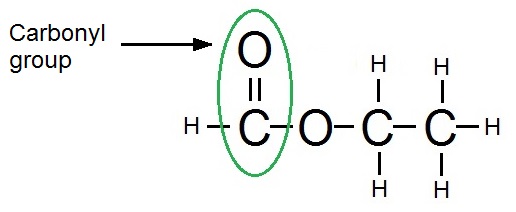
Reaction: Saponification/Hydrolysis
