Question
- Leaving Cert. Chemistry (Higher) 2017: Section B Q10
- Back to the question >
Answer
(a)
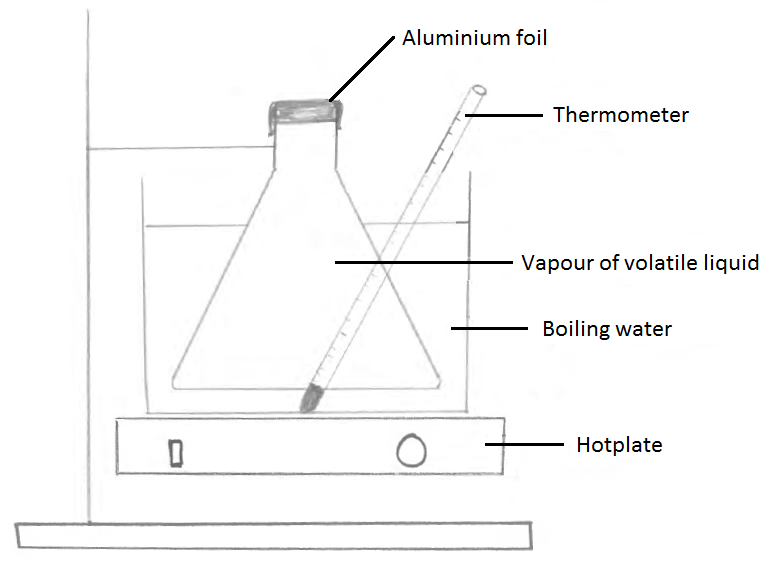
P = 101 x 103 Pa
V = 268 x 10-6 m3
R = 8.31
T = 98 + 273 = 371 K
n = PV / RT
n = 101 x 103 x 268 x 10-6 / 8.31 x 371
n = 8.779 x 10-3
(ii)
Molar Mass = 0.28 g / 8.779 x 10-3
Molar Mass = 0.28 g / 8.779 x 10-3
= 31.89 g = 32 g
